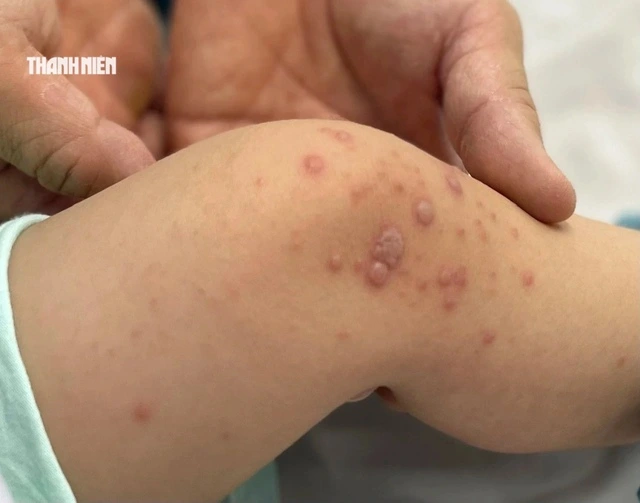
As of late March 2026, Vietnam has seen a significant spike in hand-foot-and-mouth disease (HFMD) cases, exceeding 25,000 nationwide. Medical data indicates that the number of infections has tripled since the beginning of the year, with fatalities predominantly reported in southern regions. The highly virulent EV71 strain remains the primary driver behind severe neurological complications and increased mortality rates.
In the face of the spreading outbreak, preventive measures are still limited to non-specific methods such as environmental and personal hygiene. Although the first domestic vaccine targeting the EV71 strain was granted a circulation license on March 17, 2026, the product is not yet available for public use.
The current bottleneck involves administrative pricing procedures:
-
Filing Status: As of March 31, no entities have submitted the required price declaration dossiers for this vaccine.
-
Regulatory Requirements: Under current law, price transparency is a mandatory prerequisite for pharmaceutical products to be commercially distributed following quality certification.
-
Product Specifications: The new vaccine is indicated for children aged 2 months to under 6 years, providing a 3-year protection period specifically against the most dangerous strain (EV71).
Epidemiological experts warn that the burden on the healthcare system will continue to escalate, particularly regarding severe cases, until specific immunization strategies can be officially integrated into the public health response.
Source: https://thanhnien.vn/benh-tay-chan-mieng-tang-cao-vac-xin-van-cho-cong-bo-gia-185260331202806813.htm