In a move to strengthen its oncology pipeline, pharmaceutical giant Gilead Sciences has entered into an agreement to acquire RP-3467,... Read more
Category: Uncategorized
FDA Grants First-Ever Approval for Treatment of Post-Stem Cell Transplant Microangiopathy
The U.S. Food and Drug Administration (FDA) has officially approved Yartemlea (narsoplimab), marking a historic milestone for the 31-year-old biotech... Read more
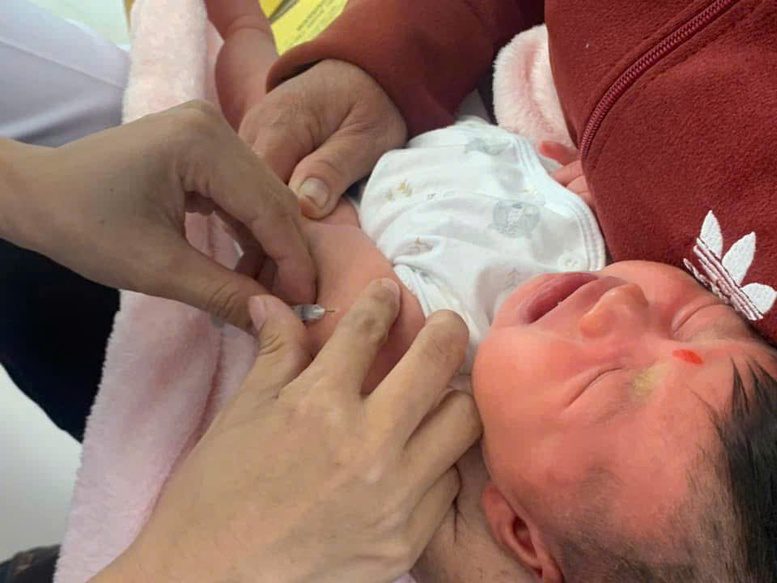
Vietnam to Provide Free Pneumococcal and HPV Vaccines Starting 2026
According to a representative from the Department of Disease Prevention (Ministry of Health) during a medical event on December 26,... Read more
Vietnam Accelerates Modernization of Pharmaceutical Regulatory Framework and Management Standards
At a recent international scientific symposium in Hanoi, regulatory authorities and healthcare experts discussed the roadmap to enhance national pharmaceutical... Read more
Healthcare Sector Proposes Strategic Shift to Combat Rising Cardiovascular Diseases
Latest medical data released during a professional conference on December 26 in Hanoi highlights a concerning public health trend. Specifically,... Read more
Medical drone delivery pilot program launched in Hanoi
Detailed Content On December 25, Duc Giang General Hospital in Hanoi officially launched a pilot program using Unmanned Aerial Vehicles... Read more
Kashiv BioSciences secures ₹648 crore financing for Gujarat manufacturing expansion
Large-scale biologics infrastructure project in Ahmedabad Kashiv BioSciences has secured 648 crore INR (approximately $77 million) in financing from the... Read more
Abbott secures FDA approval for Volt™ Pulsed Field Ablation system
Next-generation electrical pulse therapy Abbott has officially received approval from the U.S. Food and Drug Administration (FDA) for its Volt™... Read more
Aurobindo Pharma increases stake in Chinese pharmaceutical joint venture
Transaction to raise ownership to 50% Aurobindo Pharma, through its wholly-owned Dutch subsidiary Helix Healthcare B.V., has reached an agreement... Read more