National health authorities have issued an urgent nationwide recall for over 130 cosmetic items after determining that the manufacturing unit failed to maintain operational standards as required by law.
Reasons for enforcement and product scope The decision followed an on-site inspection at the factory of Vincos Vietnam Cosmetics Manufacturing JSC (located in Phu Cat, Hanoi). Despite previously holding a valid license, the facility failed to meet mandatory cosmetic production standards at the time of inspection.
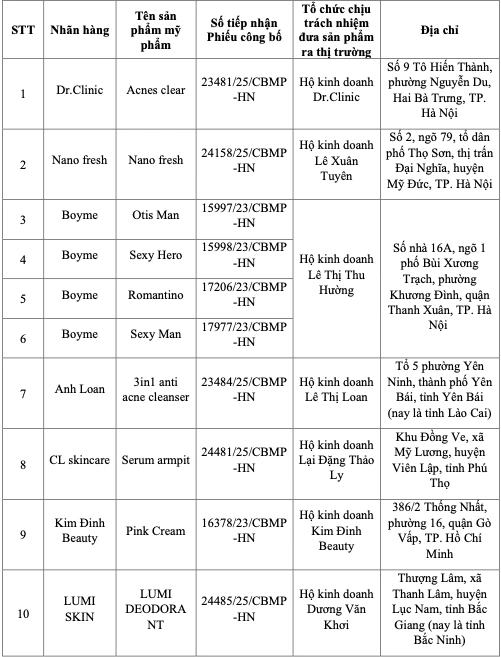
The recall list covers 131 products across various popular consumer categories, including:
-
Facial care: Cleansers, serums, toners, moisturizers, and sunscreens.
-
Body care: Shower gels, deodorants, and feminine hygiene gels.
[Image depicting regulatory documents and the list of companies responsible for the recalled products]
Remediation and oversight measures The Drug Administration of Vietnam has established a strict enforcement timeline:
-
Recall and Destruction: The manufacturer and distributing entities must notify all retail outlets, recall, and destroy all violating batches. Implementation reports must be submitted by March 3, 2026.
-
Local Supervision: Health Departments in provinces and major cities (Hanoi, HCMC, Da Nang, etc.) are tasked with monitoring the cessation of sales in their jurisdictions and reporting back by March 18, 2026.
This action is part of the health sector’s efforts to sanitize the market, ensuring that production facilities strictly adhere to safety protocols before delivering products to consumers
Source: https://vnexpress.net/131-loai-my-pham-bi-thu-hoi-tren-toan-quoc-5042113.html